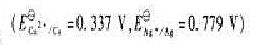题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
298k,100kpa时,用pt作电极电解sncl2水溶液,在阴极上析出sn (s),其电极电势为-0.17 v。在阳极上析出o2,已知溶液
298K,100kPa时,用Pt作电极电解SnCl2水溶液,在阴极上析出Sn (s),其电极电势为-0.17 V。在阳极上析出O2,已知溶液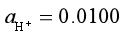 ,O2在阳极上析出的超电势为0.50 V,E?(H2O, H+?O2(g)?Pt)= 1.23 V。常数R = 8.314 J×mol-1×K-1,F = 9.65 ′ 104 C?mol-1。298 K时分解电压等于()V。精确保留至小数点后2位。
,O2在阳极上析出的超电势为0.50 V,E?(H2O, H+?O2(g)?Pt)= 1.23 V。常数R = 8.314 J×mol-1×K-1,F = 9.65 ′ 104 C?mol-1。298 K时分解电压等于()V。精确保留至小数点后2位。
 如搜索结果不匹配,请 联系老师 获取答案
如搜索结果不匹配,请 联系老师 获取答案


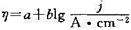 ,且Tafel常数a=0.73 V,b= 0.11 v,
,且Tafel常数a=0.73 V,b= 0.11 v,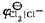 =1. 36 V,请计算实际的分解电压。
=1. 36 V,请计算实际的分解电压。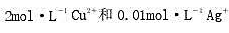 ,若以Pt为电极进行电解。
,若以Pt为电极进行电解。